Antibodies
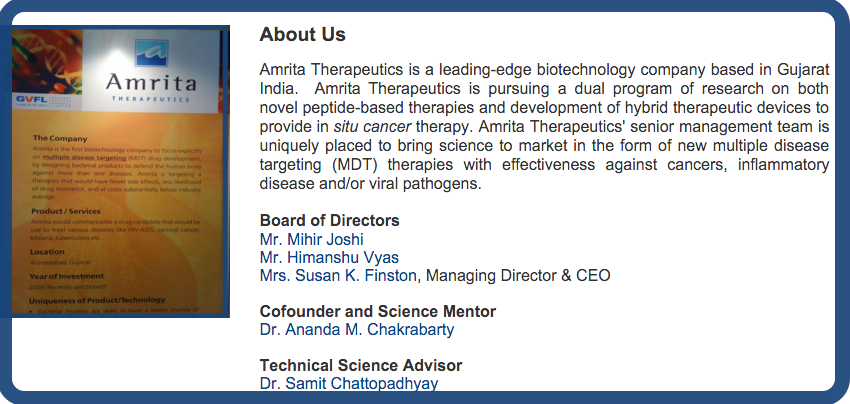
Amrita Therapeutics is in the vanguard of biotechnology companies translating microbiomic technologies into practical tools to benefit patients, with a diverse range of programs at the discovery stage and late preclinical stages.
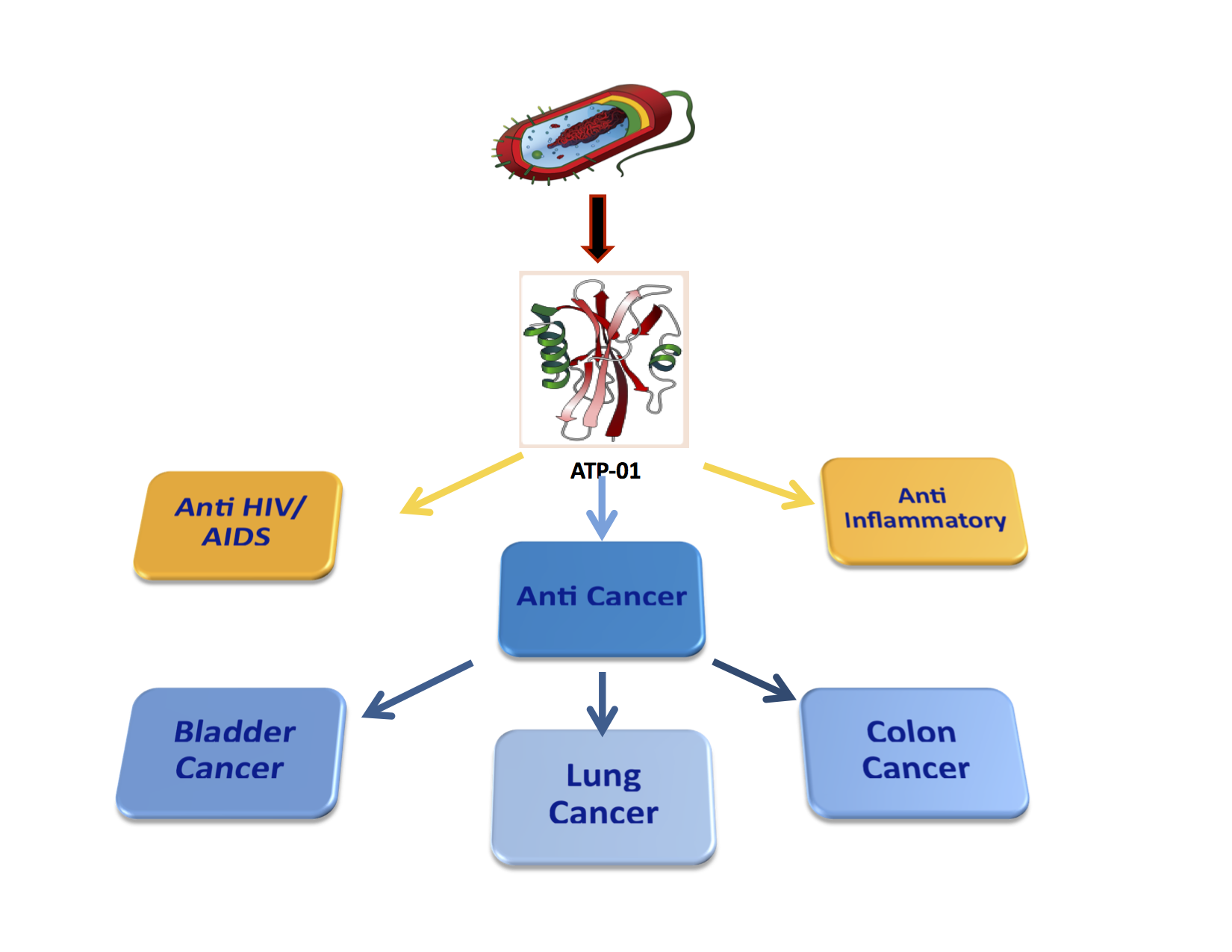
Our lead Drug Candidate AT-01C is a p53 tumor suppressor peptide derived from Mycobacterium Tuberculosis (M. bovis) Protein MPT63, also known as ATP-01.
ATP-01

AT-01C shows tumor-shrinking, anti-metastasis & anti-inflammatory activity in solid tumors including colorectal, bladder and liver cancer, binding to SMAR1 – Scaffold Matrix Attachment Region Binding Protein 1 – for down-regulation of oncogenes with little or no toxicity to healthy tissues.
In parallel to clinical development for AT-01C, Amrita Therapeutics is focusing on the 'master-regulator' SMAR1 biomarker as a companion diagnostic test to identify patients most likely to benefit from AT-01C therapy.
Amrita Therapeutics was launched at the height of the global financial crisis at Vibrant Gujarat in early 2009 with seed funding from the Gujrat Venture Finance Limited (GVFL), and has received international recognition for its innovative research platform.
Amrita Therapeutics proprietary science platform leverages our increasing understanding of the important role of the microbiome in human health, ushering in a new golden age of drug development.
Intuitive
We create easy-to-use products.
Adaptable
We build modular solutions.
Durable
We craft long-lasting goods.
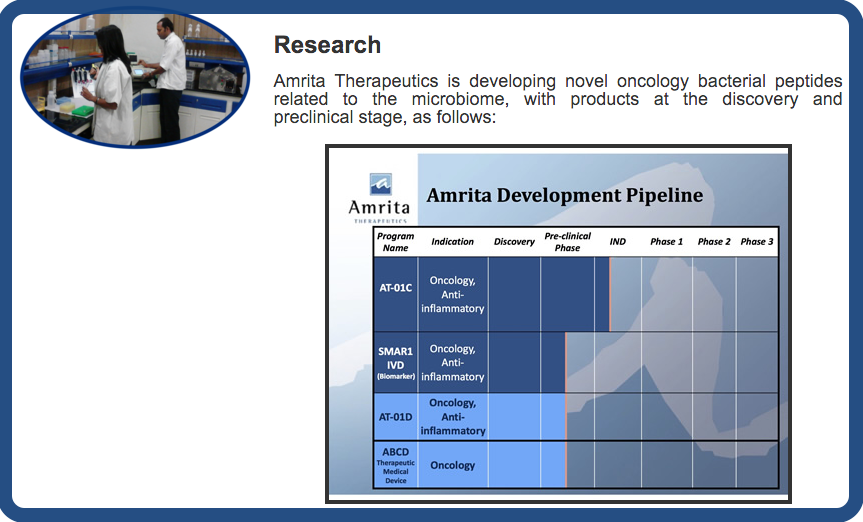
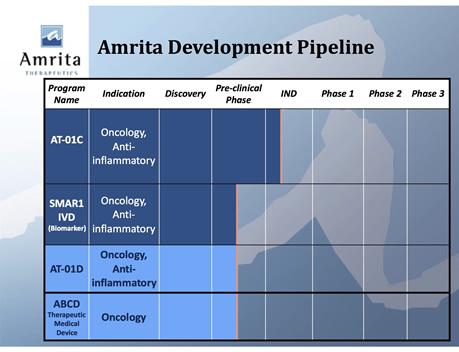
Research
Amrita Therapeutics is developing novel oncology bacterial peptides related to the microbiome, with products at the discovery and preclinical stage, as follows:
Pipeline
We have designed novel proteins and peptides including ATP-01, a protein with anticancer, anti-HIV/AIDS and anti-inflammatory activity, as well as derived truncated peptides like AT-01, a 30 amino acid peptide with activity against a range of cancers. Mechanism of Action (MOA) studies for AT-01 and its derivative peptides demonstrate both p53 (tumor suppressor) activity and anti-metastasis activity.
ATP-01
In March 2015, Amrita Therapeutics filed a provisional patent application for derived peptides from ATP-01, modified for enhanced anti-cancer activity. Amrita Therapeutics lead candidate peptide drug AT-01C upregulates p53 by stabilizing tumor ruppressor protein SMAR1 both in vitro and in vivo.
In March 2014, Amrita Therapeutics' first utility and PCT patent application relating to novel peptides for use as anti cancer therapies was published by the USPTO and EPO, available here.
In late 2013, Amrita Therapeutics initiated collaborative research on Mechanism of Action (MOA) studies for AT-01 and derivative peptides with Dr. Samit Chattopahdyay, a member of the Indian Department of Biotechnology (DBT) task force on cancer research and a recipient of the J.S. Bose National Fellow for his work on gene regulation, at the National Center for Cell Science (NCCS) in Pune, India.
In 2012 and 2013, Amrita Therapeutics filed two additional full utility and PCT patent applications for an Antiviral Compositions and Methods Thereform, and also for an innovative hybrid medical device for in situ cancer therapy.
In late 2011, Amrita Therapeutics entered into an early research compound screening program with the National Cancer Institute (NCI) in Washington, DC that demonstrated anti-cancer properties of peptide AT-01 and its derivatives.
Throughout 2011, Amrita continued and/or initiated research collaboration with apex Indian Research Institutes including the National Institute of Immunology (NII), New Delihi, India, the Centre for DNA Fingerprinting and Diagnostics (CDFD), Hyderabad, and the B.V. Patel PERD Centre, in Ahmedabad, Gujarat, and with the Instituto Superior Tecnico, in Lisbon Portugal.
In March 2010, Amrita Therapeutics was awarded a grant from the Department of Biotechnology (DBT), Ministry of Science and Technology, Government of India, under the Biotechnology Industry Partnership Program (BIPP).
Amrita Therapeutics also continues to generate candidate drugs demonstrating anti-cancer and anti-viral/parasitic activity. As noted, use of protein products of bacteria avoid the harm to patients caused by the use of the live bacteria themselves.
Publications
Our research has been highlighted in international journals including the Journal of Commercial Biotechnology (January 2012), abstract available here, and in the ISRN Neurology (2012), article vailable here.
Feel free to come up with your big idea.
We take care of the rest.
Our References
We are in good company.